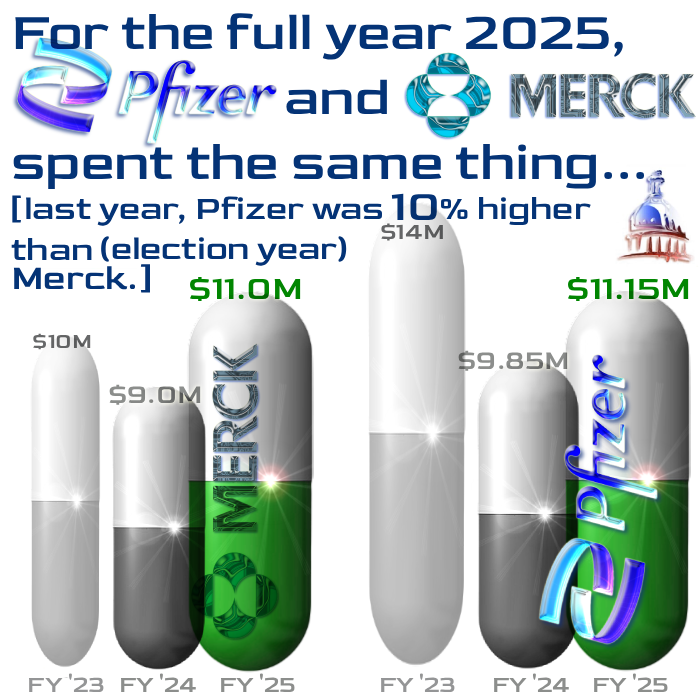
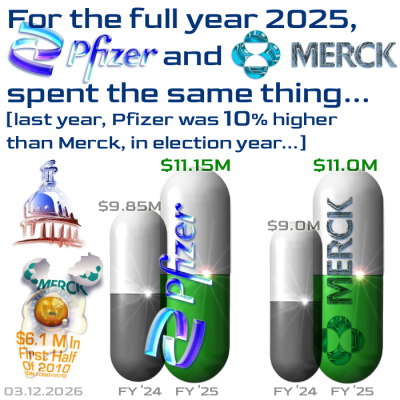
As with the others, the figures are annual, but the text describes only Q4 2025 activity — for earlier periods, just search the words “lobby spend” in the search dialogue box.
As with the others, the figures are annual, but the text describes only Q4 2025 activity — for earlier periods, just search the words “lobby spend” in the search dialogue box.
Amgen has really taken the lead dog role, in spending — right behind the overall pharma trade association, as a group (PhRMA). Here’s a sample of what they jaw-boned Congreass about — in Q4 2025:
…Issues related to drug pricing; Issues related to cardiovascular disease awareness and treatment; Issues related to FDA; Issues related to patient affordability issues, including copay cards, copay accumulators, copay maximizers, National Benefit Payment Parameters; Issues related to biosimilars reimbursement; Issues related to Supply Chain; Issues related to 340B; Issues related to pharmacy benefit managers; Issues related to biosimilars regulatory standards; Issues related to bone disease awareness and treatment; Issues related to Public law 117-1769 Inflation Reduction Act; Issues pertaining to the implementation of PL 97-414 “Orphan Drug Act,” all provisions; HR 946 Optimizing Research Progress Hope and New Cures Act; HR 1492 Ensuring Pathways to Innovative Cures Act; HR 1672 Maintaining Investments in New Innovation Act; HR 1 One Big Beautiful Bill Act (An Act to provide for reconciliation pursuant to title II of H. Con. Res. 14); S 1954 Biosimilar Red Tape Elimination Act; HR 5526 Biosimilar Red Tape Elimination Act; HR 2214 DRUG Act; H.R.1968 – Full-Year Continuing Appropriations and Extensions Act, 2025; HR 5509 Safe Step Act; HR 5256 340B Access Act; HR 4581 340B Patients Act; S 2296 NDAA FY2026 National Defense Authorization Act, issued related to Biosecure Act; H.R. 6703 Lower Health Care Premiums for All Americans Act; S 3345 PBM Price Transparency and Accountability Act; H.R. 6166 Lowering Drug Costs for American Families Act; S.3019 No Big Blockbuster Bailouts Act; H.Res. 928 Affirming support for most-favored-Nation drug pricing for United States patients
S 3349 PBM Disclosure Act; HR 4317 PBM Reform Act; S 3510 Biosimilar Inspection Modernization Act….
Issues related to drug pricing; Issues related to Public law 117-1769 Inflation Reduction Act; Issues related to cardiovascular disease awareness and treatment; Issues related to reimbursement for biologics/biosimilars; Issues related to PBM reform; Issues related to bone disease awareness and treatment — HR 946 Optimizing Research Progress Hope and New Cures Act; HR 1492 Ensuring Pathways to Innovative Cures Act; HR 1672 Maintaining Investments in New Innovation Act; HR 1 One Big Beautiful Bill Act (An Act to provide for reconciliation pursuant to title II of H. Con. Res. 14); H.R.1968 – Full-Year Continuing; Appropriations and Extensions Act, 2025; S.3345, PBM Price Transparency and Accountability Act; H.R. 6166, Lowering Drug Costs for American Families Act; S.3019 – No Big Blockbuster Bailouts Act; H.Res. 928 – Affirming support for most-favored-Nation drug pricing for United States patients; S 3349 PBM Disclosure Act; HR 4317 PBM Reform Act….
Issues related to corporate and international tax, including regarding Public Law 115-97, Tax Cuts and Jobs Act; Issues related to Puerto Rico; Issues related to OECD negotiations on the taxation of global income; Public Law 119-21, One Big Beautiful Bill Act (An Act to provide for reconciliation pursuant to title II of H. Con. Res. 14); Issues related to patents and taxes….
Federal Trade Commission related issues, no specific bill; Issues related to the Patent Act, no specific bill; Issues related to March-In/Bayh Dole, WTO/TRIPS waiver, no specific bill; Issues related to patent thickets/product hopping; Issues related to obviousness/ double patenting; Issues related to FDA/PTO coordination; Issues related to skinny labeling; Issues related to patents and taxes; S.1041 A bill to amend title 35, United States Code, to address the infringement of patents that claim biological products, and for other purposes; S.1040 — A bill to amend the Federal Trade Commission Act to prohibit product hopping, and for other purposes; S.2296– NDAA (FY 2026 National Defense Authorization Act — BIOSECURE language); S 708/ HR 1574 — Realizing Engineering, Science and Technology Opportunities by Restoring Exclusive Patent Rights Act of 2025 (RESTORE); S.1553/HR3160 — Promoting and Respecting Economically Viable American Innovation Act (PREVAIL); S. 1546/ HR 3152– Patent Eligibility Restoration Act of 2025 (PERA); S 2276 / HR 3269 Eliminating Thickets to Increase Competition Act (ETHIC Act); HR 6485 Skinny Labels Big Saving Act….
There you have it. Onward — with big winds — and snow, ahead! Grin….
नमस्ते